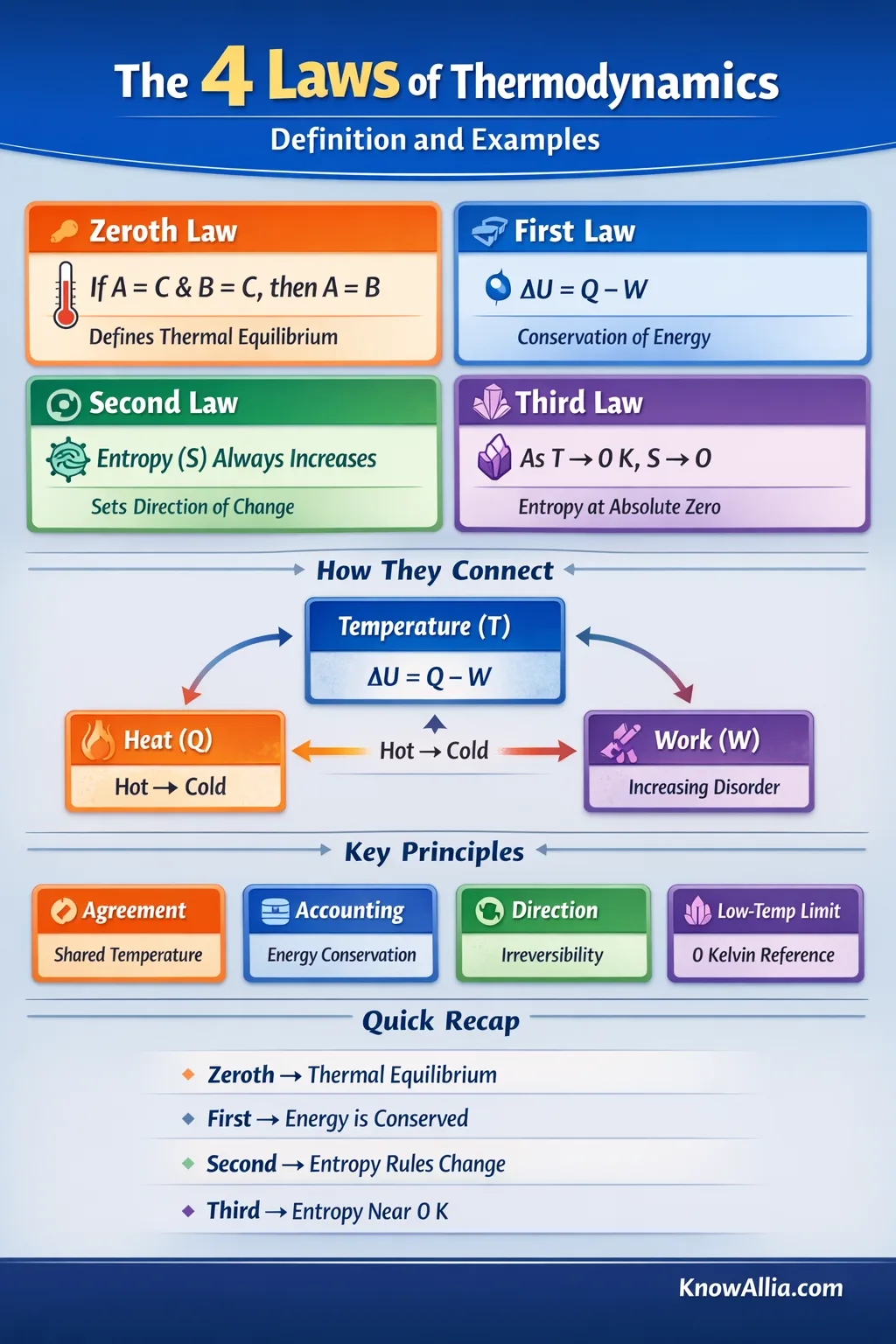
The four laws of thermodynamics are the basic rules that connect temperature, heat, work, and entropy. They explain why thermometers can agree, why energy accounting always balances, why some changes have a clear “direction,” and why absolute zero is a limit you can approach but not reach in practice.[g]↗
A Clear Starting Point
Thermodynamics is about energy moving and energy changing form in systems you can measure—engines, refrigerators, living rooms, batteries, oceans, and more. The laws don’t tell you how fast something happens; they tell you what is allowed, what is not, and what trade-offs come with real-world processes.
- Zeroth Law: makes temperature a consistent idea.
- First Law: keeps the energy ledger balanced.
- Second Law: sets the direction of natural change.
You’ll learn what each law means in plain English, the key words people mix up, and the kinds of everyday situations each law explains—without turning the topic into a “tips” article.
What Thermodynamics Covers
Thermodynamics is a big-picture way to describe physical systems. Instead of tracking every molecule, it tracks measurable quantities—like temperature, pressure, volume, and entropy—and relates them through the laws.[c]↗
What These Laws Do (and Don’t) Say
- They describe possible vs. impossible changes, not how quickly they happen.
- They apply to many systems, but they usually assume the system is described well by macroscopic properties.
- They don’t replace chemistry, fluid dynamics, or quantum mechanics; they give a constraint layer those fields must respect.
Zeroth Law: Why Temperature Is a “Shareable” Fact
The zeroth law says: if system A is in thermal equilibrium with system C, and system B is also in thermal equilibrium with system C, then A and B are in thermal equilibrium with each other. That simple idea is what makes temperature meaningful and measurable with a thermometer.[a]↗
- Plain-English Meaning
- “No net heat flow” is a consistent relationship you can compare across objects.
- What It Enables
- Thermometers can be calibrated and trusted across different materials.
Example: You place a thermometer in a glass of water. After a short wait, the reading settles. That “settled” moment is the thermometer and water reaching thermal equilibrium, so the thermometer’s temperature matches the water’s temperature.[n]↗
First Law: Energy Accounting Always Balances
The first law of thermodynamics is energy conservation for heat-and-work problems. One common form is: the change in a system’s internal energy equals the heat added to the system minus the work done by the system.[b]↗
ΔU = Q − W (common physics sign convention: W is work done by the system)You’ll also see another perfectly valid form in chemistry: ΔU = Q + W, where W is defined as work done on the system. The physics doesn’t change—only the bookkeeping sign does.[j]↗
An Analogy That Usually Clicks
Think of internal energy like your bank balance. Heat and work are the ways money moves in or out. You can spend money in different ways, but the ledger still has to add up. The first law is that ledger: it won’t let you “create” energy out of nowhere, even if the path looks clever.
- Example (heating a pot): Put a covered pot of water on a stove. Heat flows in (Q). Some energy raises the water’s internal energy (ΔU), and some can go into expansion work (W) as the contents warm.
- Example (compressed air): Rapidly compressing air warms it. That’s work done on the gas showing up as a higher temperature and higher internal energy.
Second Law: Why Some Changes Have a Direction
The second law of thermodynamics introduces entropy and explains why real processes aren’t perfectly reversible. A practical way to say it: for an isolated system, total entropy does not decrease. In everyday life, that shows up as heat flowing naturally from hot to cold, and as a limit on how efficiently heat can be turned into work.[c]↗
What Entropy Is Doing Here
Entropy can be described in more than one way depending on context, but a safe starting point is: it’s a state variable that helps track which energy changes are possible and which are not, especially when heat is involved.[d]↗
For a reversible heat transfer step: ΔS = Qrev / TEveryday example: A hot mug cools down in a room. The first law allows many energy exchanges, but the second law explains why you don’t see the reverse—room air spontaneously heating the mug while the room gets colder overall—unless you add outside work (like a heat pump).
Common Places the Second Law Shows Up
- Heat engines: you can convert heat to work, but not all of it in a cycle.
- Mixing: cream spreads through coffee without you stirring, but “unmixing” doesn’t happen on its own.
- Friction: ordered motion (like spinning) easily turns into random thermal motion, not the other way around.
One more detail that matters: the second law is often described as statistical. It’s not that reversals are “forbidden by magic”; it’s that, for the huge number of particles in everyday systems, the reverse is so unlikely that it effectively never happens in real time. University-level notes often explain this “probability and direction” link in more depth.[i]↗
Third Law: The Low-Temperature “Zero Point” for Entropy
The third law of thermodynamics gives entropy a reference point near absolute zero. A widely used statement is: the entropy of an ideally ordered (perfect) crystal approaches zero as temperature approaches 0 K. That reference makes it possible to talk about absolute entropy, not just entropy changes.[d]↗
Real materials can have defects or “frozen-in” disorder, so the perfect-crystal picture is an ideal limit. Educational chemistry references often use the third law to explain why a clean entropy scale can be built from low temperatures upward.[h]↗
The Four Laws Side by Side
Here’s a compact comparison. Notice how each law answers a different kind of question: agreement (zeroth), accounting (first), direction (second), and low-temperature reference (third).
| Law | Core Idea | What It Lets You Do | Simple Example |
|---|---|---|---|
| Zeroth | Thermal equilibrium is consistent and transitive. | Define and measure temperature reliably. | Thermometers can be calibrated and can agree across materials. |
| First | Energy is conserved in heat/work processes. | Track energy flows with Q, W, and ΔU. | Compressing gas warms it; expansion can cool it. |
| Second | Entropy helps set the direction of natural change. | Understand why 100% heat-to-work cycles fail and why mixing happens. | Hot coffee cools in a room; it doesn’t heat up by itself. |
| Third | Entropy approaches a constant as T → 0 K. | Use an absolute reference for entropy near absolute zero. | Low-temperature behavior and absolute entropies become definable. |
Infographic: How the Laws Connect in One Picture
Four Laws, One Connected System
Thermal equilibrium defines temperature, energy conservation balances the books, entropy sets direction, and the low-temperature limit anchors the entropy scale.
How Heat, Work, and Entropy Relate
Temperature (T) Anchored by equilibrium Heat Transfer (Q) Energy crossing due to ΔT Internal Energy (U) State of the system Work (W) Energy via forces & motion Entropy (S) Direction & reversibility First law: ΔU balances Q and W Second law: S tracks direction A conceptual map (not to scale)What Each Law Controls
Thermal equilibrium makes temperature comparisons consistent.
Energy bookkeeping: heat and work change a system’s internal energy.
Entropy gives a direction to real processes and limits efficiency.
The entropy scale has a low-temperature reference near 0 K.
Zeroth → Temperature
Why thermometers can agree after contact.
First → Conservation
Why you can’t get “free energy” from a cycle.
Second → Direction
Why heat engines have a ceiling and mixing happens.
Third → Low-Temp Limit
Why entropy has a clean reference near absolute zero.
Key Terms (Mini Glossary)
- System
- The part of the world you’re analyzing; everything else is the surroundings.
- State Variable
- A property like T, U, or S that depends only on the current state, not the path taken.
- Heat (Q)
- Energy transferred because of a temperature difference, not “stored heat.”
- Work (W)
- Energy transferred via forces acting through distance (like expansion against pressure).
- Internal Energy (U)
- Energy associated with the system’s state (microscopic motion and interactions, in aggregate).
- Entropy (S)
- A state variable tied to reversible heat transfer and the direction of spontaneous change.[d]↗
Common Misconceptions and Where Confusion Starts
- “Heat is something stored inside objects.” In thermodynamics, heat is typically treated as energy in transit due to a temperature difference, not a substance sitting inside matter.
- “Entropy just means disorder.” “Disorder” can be a helpful mental picture in some cases, but entropy is more dependable as a state variable tied to reversible heat transfer and the direction of processes.[c]↗
- “The second law says energy is wasted.” Energy is still conserved (first law). The second law is about usefulness: some energy ends up spread out in ways that are harder to turn back into organized work.
- “Absolute zero is just a very cold place.” It’s a defined limit of the temperature scale; the third law is one reason reaching it exactly is not something you can do in a finite, practical procedure.
- “The laws only matter for engines.” Engines are a classic example, but the same ideas show up in everyday cooling, phase changes, electronics heating, weather, and materials behavior.
Limitations and What We Don’t Know From the Laws Alone
The thermodynamics laws are incredibly reliable, but they don’t answer every question by themselves. They won’t tell you rates (how fast heat moves), and they don’t automatically predict the detailed path a complex system will take—only what outcomes are compatible with energy conservation and entropy constraints. For many real problems, you combine thermodynamics with heat transfer, fluid flow, chemistry, or materials data.[i]↗
Where Care Is Needed
- Sign conventions: whether you write Q − W or Q + W depends on the definition of W.[j]↗
- Ideal limits: statements like “entropy is zero at 0 K” assume an ideal perfectly ordered crystal.[h]↗
- Practical temperature realization: measurement standards evolve; modern definitions tie temperature to fundamental constants, not a single reference sample.[e]↗
FAQ
Questions People Ask (and the Straight Answers)
Are there really four laws of thermodynamics?
Yes. People often say “the three laws,” because the zeroth law was named later, but today the standard set is zeroth, first, second, and third.[g]↗
Does the first law mean you can always get useful work out?
No. The first law says energy is conserved. The second law adds that not all energy is equally convertible into work in real processes, especially when heat is involved.[c]↗
Can entropy ever decrease?
In a local part of a system, entropy can go down if something else pushes it that way (for example, by doing work or exporting entropy to the surroundings). The key second-law idea is about the total for an isolated system not decreasing.[c]↗
What exactly is the kelvin based on today?
The kelvin is defined by fixing the numerical value of the Boltzmann constant k to 1.380 649 × 10−23 J K−1. That ties thermodynamic temperature directly to energy at the molecular scale.[e]↗
Does the third law say you can reach absolute zero if you try hard enough?
No. The third law is often taught as a limit statement about entropy as temperature approaches 0 K. In practice, it supports the idea that reaching 0 K exactly is not something you can do in a finite, real procedure, even though you can get very close.[h]↗
Sources
- [a]↩ NASA Glenn Research Center – Zeroth Law: Thermal Equilibrium (Used for the zeroth law definition and thermometer logic.)
- [b]↩ NASA Glenn Research Center – First Law: Internal Energy (Used for the first law statement and the Q/W framing.)
- [c]↩ NASA Glenn Research Center – Second Law: Entropy (Used for the second law and the reversible entropy-change relation.)
- [d]↩ IUPAC Gold Book – “Entropy” (E02149) (Used for the entropy definition and the 0 K perfect-crystal reference.)
- [e]↩ BIPM – SI Base Unit: Kelvin (K) (Used for the modern definition of kelvin via the Boltzmann constant.)
- [f]↩ NIST – Kelvin: Boltzmann Constant (Used to confirm the SI redefinition context and why k is central.)
- [g]↩ Encyclopaedia Britannica – Laws of Thermodynamics (Used for the general framing of “four laws” and historical naming context.)
- [h]↩ Purdue University – Entropy and the 2nd & 3rd Laws of Thermodynamics (Used for the third-law perfect-crystal explanation and entropy-scale framing.)
- [i]↩ MIT OpenCourseWare – Thermodynamics of Materials (Lecture Notes) (Used as a higher-detail reference on how the laws are used in real thermodynamic analysis.)
- [j]↩ Georgia State University – HyperPhysics: First Law of Thermodynamics (Used to confirm the common sign-convention difference between physics and chemistry forms.)
- [n]↩ NIST – Kelvin: Present Realization (Used for the practical description of reaching thermal equilibrium when calibrating temperature measurements.)
If you keep just one idea in mind, make it this: the laws don’t try to describe every detail of nature—yet they still put hard boundaries on what any real process can do with heat, work, and temperature.