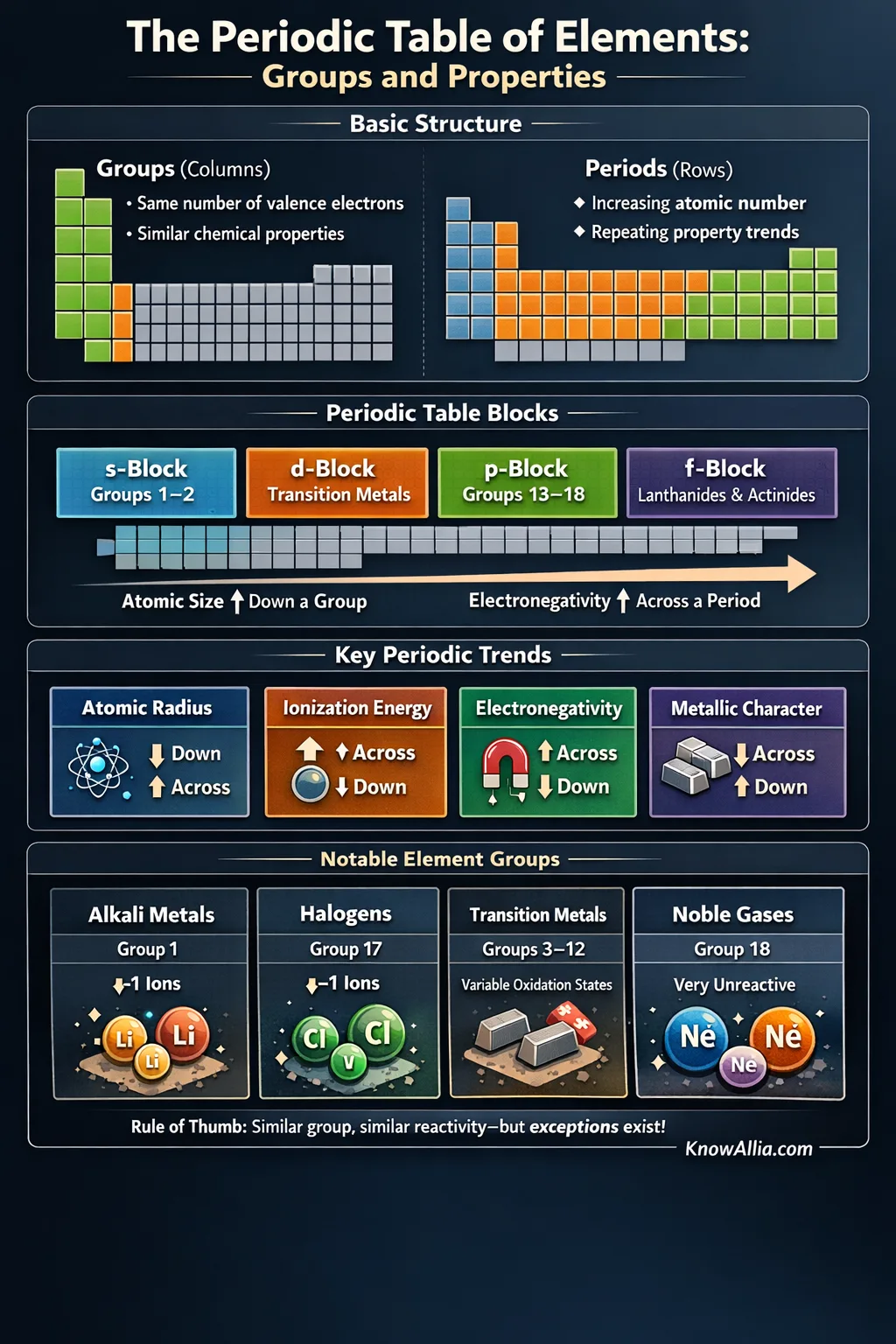
The periodic table is a map of all known chemical elements arranged by atomic number (the number of protons). Its layout is designed so elements with similar behavior line up in vertical columns called groups, while horizontal rows called periods reflect repeating patterns in electron structure.[e] 🔗
A Clean Overview Before We Go Deeper
When people say “groups and properties,” they usually mean one idea: elements in the same column tend to react in similar ways because their outer electrons follow the same pattern. The table also helps you predict how properties shift as you move across or down.
- Groups (columns) connect elements with related chemistry.
- Blocks (s, p, d, f) describe which electron “region” is being filled.
- Many trends are real, but exceptions are part of the story.
You’ll come away knowing how groups are numbered, why certain families (like halogens and noble gases) act the way they do, how blocks relate to electrons, and where the table still has honest gray areas rather than perfect rules.
Table Basics
Modern periodic tables list elements in order of increasing atomic number, which naturally groups similar chemistry into columns.[e] 🔗 Each element’s “cell” often shows a symbol, atomic number, and an atomic weight value. If you want a set of values that’s carefully reviewed and maintained, organizations like NIST publish periodic-table data aimed at accuracy and consistency.[b] 🔗
What A “Group” Really Means
A group is a vertical column. Elements in the same group often share the same count of valence electrons (outer electrons), which heavily influences bonding and reactivity.
What A “Period” Tells You
A period is a horizontal row. Moving left to right, you add protons and electrons, and the outer-electron pattern changes in a way that makes properties repeat in a recognizable rhythm.
One easy analogy: think of the periodic table like an apartment building. Groups are vertical stacks where apartments share the same layout, while periods are floors where the layout gradually changes from one end to the other. The “view” outside the window changes, but the building’s blueprint still repeats.
Groups and Why They Share Properties
Today, the most widely used group labels are simply 1 through 18. The International Union of Pure and Applied Chemistry recommends this numbering system, which avoids older schemes that can be inconsistent across countries or textbooks.[a] 🔗
In the main-group elements (mostly groups 1–2 and 13–18), group similarity is often strong because outer-electron patterns line up neatly. In the transition metals (groups 3–12), you still see family resemblances, but they can be subtler because d-electrons add extra options for bonding.
- Same group usually means a related “outer electron” pattern.
- That pattern often leads to familiar ion charges (like +1, +2, or −1) in many common compounds.
- Down a group, atoms generally get larger, which can shift how strongly electrons are held.
Small but important detail: “Similar” does not mean “identical.” Even within one group, the balance between size, electron shielding, and bonding options changes as you go down.
Periods, Blocks, and Electron Structure
Alongside groups and periods, you’ll often hear about blocks: s, p, d, and f. Blocks describe which type of electron “slot” is being filled as you move across the table, and many periodic tables explicitly label these sections.[d] 🔗
- s-block
- Mostly groups 1–2 (plus helium). Often strongly “group-like” behavior.
- p-block
- Groups 13–18. Includes many familiar nonmetals and metalloids.
- d-block
- Groups 3–12. The transition metals, known for flexible bonding.
- f-block
- The lanthanoids and actinoids, usually shown below to keep the table compact.
Periodic Trends That Actually Show Up
Periodic trends are not magic; they’re the table’s structure showing its hand. As you move across a period, the nucleus gains protons, and that tends to pull electrons in more strongly. As you move down a group, new electron shells add distance and shielding. Courses and reference materials often summarize this using familiar trends like atomic radius, ionization energy, electron affinity, and electronegativity.[f] 🔗
| Property | Across A Period (Left → Right) | Down A Group (Top → Bottom) | Why It Tends To Happen |
|---|---|---|---|
| Atomic Radius | Often decreases | Often increases | More nuclear pull across; more shells down |
| Ionization Energy | Often increases | Often decreases | Electrons held tighter across; farther away down |
| Electronegativity | Often increases | Often decreases | Atoms attract bonding electrons more across; less down |
| Metallic Character | Often decreases | Often increases | Metals more common toward lower-left of the table |
Reality check: trends are most reliable when you compare nearby elements and use the same measurement basis. Some values depend on definition, experimental method, or the chemical environment, especially for heavier elements and transition metals.
Group Snapshots You’ll See Most Often
Many group labels have common “family” names that show up in textbooks, charts, and digital tables. Interactive periodic tables typically make these group boundaries easy to see, including group numbers and blocks.[d] 🔗
| Group(s) | Common Name | Typical Outer Pattern | Common Chemistry Signals |
|---|---|---|---|
| 1 | Alkali Metals | Often ends in ns1 | Frequently form +1 ions; many react readily with other elements |
| 2 | Alkaline Earth Metals | Often ends in ns2 | Commonly form +2 ions; often make stable salts and oxides |
| 3–12 | Transition Metals | d-electrons involved | Variable oxidation states; many are strong conductors and useful catalysts |
| 13 | Boron Group | Often ns2np1 | Many compounds trend toward +3, with notable differences down the group |
| 14 | Carbon Group | Often ns2np2 | Ranges from nonmetal to metal down the group; common covalent bonding |
| 15 | Nitrogen Group | Often ns2np3 | Can show multiple oxidation states; many form important oxides and acids |
| 16 | Chalcogens (Oxygen Group) | Often ns2np4 | Many form −2 ions; key in oxides and sulfides |
| 17 | Halogens | Often ns2np5 | Often form −1 ions; strong tendency to make salts |
| 18 | Noble Gases | Closed shell (He: 1s2) | Commonly low reactivity; often gases at room conditions[e] 🔗 |
If you’re checking numbers like “atomic weight,” it helps to know what that value represents. The Commission on Isotopic Abundances and Atomic Weights (CIAAW) publishes standard atomic weights meant to apply to normal materials, and it also explains why some elements carry more uncertainty than others.[c] 🔗
How Groups, Periods, and Blocks Fit Together
The table’s shape is not decorative. The left and right edges hold most “main-group” patterns, the middle carries transition-metal variety, and the two lower rows keep the layout compact while preserving the underlying structure.
Block Layout (Simplified)
Elements align by related outer-electron patterns, so families often share typical charges and bonding styles.
As you move left to right, the outer shell fills in a way that makes properties repeat in a recognizable cycle.
The block tells you which electron region is being filled, which helps explain why chemistry shifts across the table.
Main-Group Reliability
Group similarity is usually strongest in the s- and p-block because the outer pattern lines up cleanly.
Transition-Metal Variety
d-electrons can participate in bonding in multiple ways, so oxidation states and properties can be more flexible.
Why Atomic Weights Vary
Some “weights” depend on natural isotope mixtures, so authoritative tables explain uncertainty and intervals.
Common Misconceptions and Confusions
- “Group numbers are universal everywhere.” Older labels (like A/B systems) can disagree with each other. The clean 1–18 system is meant to reduce that confusion.[a] 🔗
- “Every element in a group behaves the same.” The family resemblance is real, but size and electron shielding change down a column, and transition metals add extra complexity.
- “Atomic weight is a single, fixed number.” For some elements, natural isotope variation means a range is more honest than a single value, which is why standard tables discuss uncertainty categories.[c] 🔗
- “The f-block isn’t part of the table.” It’s part of the structure; it’s often displayed separately to keep the main table compact.[e] 🔗
Key Terms
- Atomic Number
- The number of protons in an atom’s nucleus; it sets the element’s identity.[e] 🔗
- Group
- A vertical column (1–18) where elements often share similar outer-electron patterns and related chemistry.[a] 🔗
- Period
- A horizontal row; moving across changes how the outer shell fills, creating repeating patterns.
- Block (s, p, d, f)
- A section of the table tied to which electron region is filling; many periodic tables label these explicitly.[d] 🔗
- Valence Electrons
- Outer-shell electrons that most strongly influence bonding and reactivity.
- Ionization Energy
- The energy needed to remove an electron; a key trend often discussed alongside radius and electronegativity.[f] 🔗
- Standard Atomic Weight
- A recommended value intended to apply to normal materials, with uncertainty explained by isotopes and measurement limits.[c] 🔗
Where the Table Still Has Gray Areas
The periodic table is one of science’s most useful organizing tools, but it’s not a perfect set of hard borders. Some questions remain open or depend on conventions rather than a single “forever answer.”
- Group 3 placement has been debated: some layouts place La/Ac, others Lu/Lr, and IUPAC has noted the question explicitly.[a] 🔗
- Hydrogen’s “family” is famously awkward; it shares traits with more than one group depending on context, so it’s often treated as special rather than perfectly “matched.”
- Trend exceptions are normal, especially when comparing elements far apart or when different measurement definitions are used for the same property.[f] 🔗
- Atomic-weight precision can be limited by natural isotope variation, not just lab measurement, so some elements are better represented by intervals or carefully qualified values.[c] 🔗
None of these gray areas undermine the table’s value. They’re simply reminders that the periodic table is a human-made layout built to reflect nature as clearly as possible—while staying practical to read and use.
FAQ
Answers People Usually Look For
Why do elements in the same group behave similarly?
In many cases, elements in the same group share a related outer-electron pattern. Since those outer electrons are the ones most involved in bonding, similar patterns often lead to similar reactivity and common ion charges.
What does “group 1–18” mean, and who decided that?
It’s the modern numbering system for the table’s columns. IUPAC recommends numbering groups from 1 to 18 to avoid confusion from older labeling systems that don’t always match each other.
Are blocks (s, p, d, f) the same thing as groups?
No. Groups are columns. Blocks are bigger regions of the table tied to which electron region is filling. Blocks help explain why properties shift across a row and why the table has its familiar shape.
Why are the lanthanoids and actinoids shown at the bottom?
Mostly for readability. Many tables keep an 18-column layout by placing the f-block as two separate rows below, even though those elements are part of the same overall structure.
Is atomic weight a constant value for each element?
Not always. Some elements vary in natural isotopic composition, so authoritative sources may describe uncertainty categories or even provide intervals to reflect real-world materials.
Do periodic trends work everywhere on the table?
They work best as patterns, not rigid laws. Trends like radius, ionization energy, and electronegativity are most dependable when comparing nearby elements and using consistent definitions and measurement approaches.
Sources
These references were chosen for editorial rigor and long-term reliability. Each link is used only once in this article, and the letter tags connect citations to their matching source entry.
- [a] ↩ IUPAC – Periodic Table of Elements (Group numbering 1–18, collective series names, and the noted group 3 placement question.)
- [b] ↩ NIST – Periodic Table of the Elements (Critically evaluated atomic property data and an official printable table.)
- [c] ↩ CIAAW – Standard Atomic Weights (How standard atomic weights are defined and why uncertainty differs by element.)
- [d] ↩ Royal Society of Chemistry – Periodic Table (Clear presentation of groups, periods, and s/p/d/f blocks in a modern table layout.)
- [e] ↩ Encyclopaedia Britannica – The Periodic Table (Overview of organization by atomic number, periods, and common structural conventions.)
- [f] ↩ MIT OpenCourseWare – Periodic Trends (Lecture Notes) (Solid explanations of trends like ionization energy, electron affinity, electronegativity, and radius.)