The states of matter are the main physical forms a substance can take—solid, liquid, gas, and plasma—based on how tightly its particles are held together and how freely they move.[a]🔗
A Clear Way to Think About It
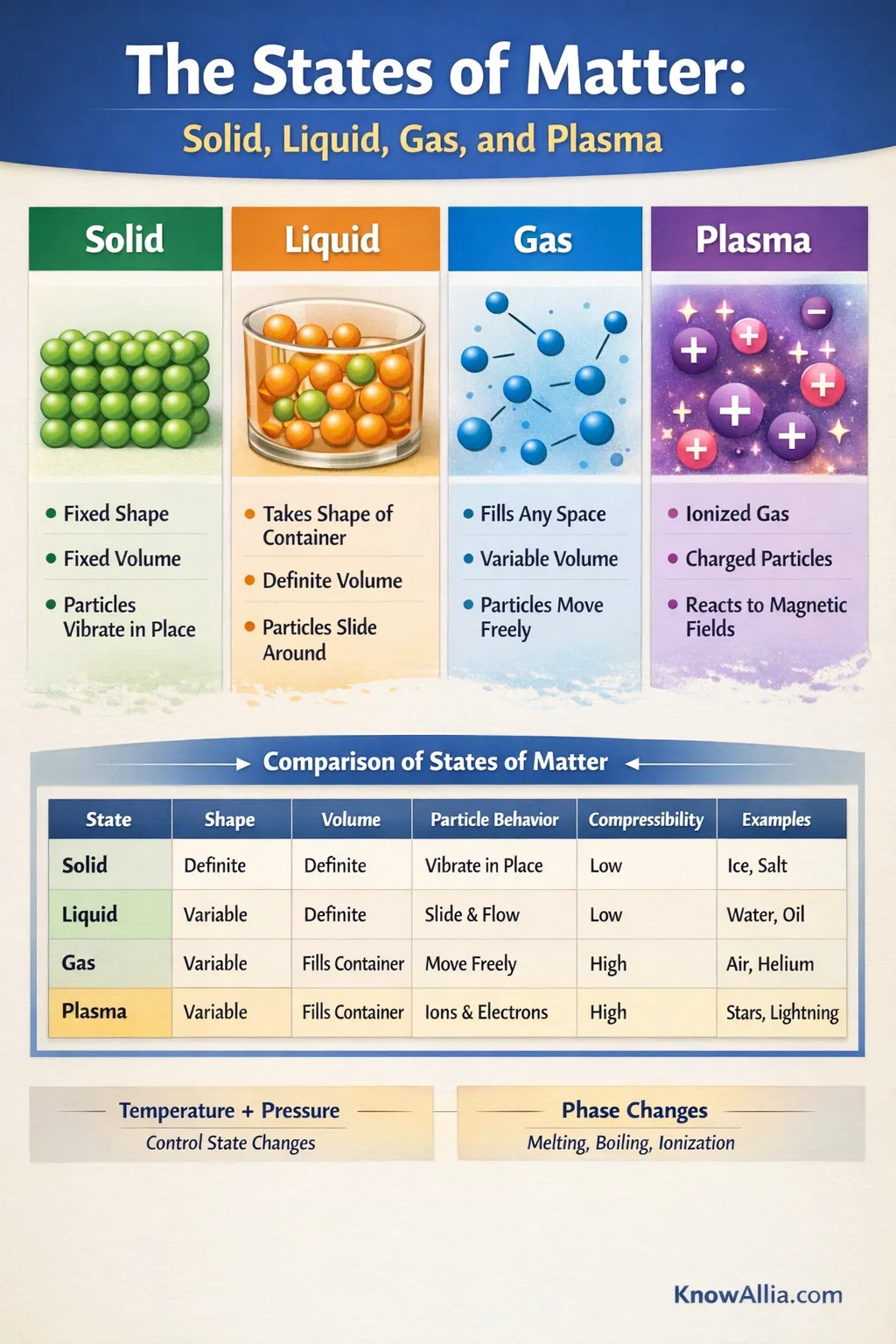
Solids keep their shape, liquids keep their volume, gases expand to fill space, and plasma behaves like a gas with charged particles that react strongly to electric and magnetic fields.[a]🔗 The “right” state is mainly controlled by temperature and pressure, not temperature alone.[c]🔗
- Particle freedom rises from solid → liquid → gas → plasma
- Phase changes often “spend” energy on rearranging particles, not raising temperature
- Real life includes edge cases (glass, supercritical fluids), so boundaries can blur
What you’ll learn here: how each state behaves, what’s happening at the particle level, why pressure matters so much, and how to avoid the most common mix-ups (like vapor vs gas, or plasma vs “just hot gas”).
On This Page
What Makes a State of Matter?
A “state” is basically a behavior package: how a substance holds shape and volume, how it flows, and how easily it compresses.[a]🔗 Those behaviors come from a tug-of-war between particle motion (thermal energy) and the forces pulling particles together.
Here’s a helpful analogy: imagine a crowded concert. In a solid, people are packed in fixed spots (they can wiggle, but not swap places). In a liquid, they’re still close, but they can slide past one another. In a gas, the crowd spreads out to fill the whole venue. Plasma is like the gas crowd—except many people are carrying charged balloons that make them react to invisible “field” rules, changing how the whole crowd moves together.
What You Can Observe
- Shape: does it keep a form or take the container’s shape?
- Volume: does it stay the same or expand to fill space?
- Compressibility: can you squeeze it smaller easily?
- Flow: does it pour, spread, or resist motion?
What’s Happening Inside
- Spacing: how far particles are from each other
- Freedom: whether particles mainly vibrate or also move around
- Order: regular patterns (crystals) vs messy arrangements (glasses)
- Charge: free electrons/ions that make plasma special
Solid: Order, Rigidity, and Two Common “Flavors”
A solid keeps a definite shape and definite volume because its particles are locked into place and mostly vibrate instead of traveling around.[a]🔗 Solids usually resist compression because there’s not much empty space to squeeze out.
Not all solids are the same on the inside. A practical way to sort them is by atomic-level order:
- Crystalline solids have repeating patterns (think table salt or a snowflake’s structure).
- Amorphous solids (often called glass) lack long-range order, even though they feel rigid in your hand.[g]🔗
Example you can picture: window glass doesn’t have the neat repeating “grid” of a crystal, but it still behaves like a solid at everyday timescales. That “messy but rigid” structure is why it’s often used as the go-to example of an amorphous solid.[g]🔗
Liquid: Flow With a Fixed Volume
A liquid has a definite volume but no fixed shape—it flows to match its container.[a]🔗 Its particles stay close together, yet they can rearrange, which is why you can pour it.
Liquids also have some “signature” properties that make them feel different from gases:
- Viscosity: how strongly a liquid resists flowing (honey vs water).
- Surface tension: the “skin-like” effect at the surface that helps droplets form.
- Slow diffusion: smells spread far faster in air than dissolved color spreads through still water.
People often say liquids are “incompressible.” That’s mostly true in everyday life, but it’s better to say liquids are hard to compress, not impossible to compress.
Gas: Expand, Mix, and Compress
A gas has neither a fixed shape nor a fixed volume—it expands to fill its container.[a]🔗 Gas particles are far apart and move freely, which is why gases compress easily and why odors spread quickly through a room.
A detail many explanations skip: the words gas and vapor can be used differently in science. The key idea is the critical temperature—at and above it, the gaseous form can’t be liquefied by pressure alone.[h]🔗 Below that temperature, a gaseous form is often called a vapor because it can be condensed by squeezing it enough at the same temperature.
Plasma: Ionized Matter That “Listens” to Fields
Plasma is a gas that’s at least partly ionized, meaning it contains free electrons and ions, while staying roughly electrically neutral overall.[e]🔗 That mix of charged particles makes plasma electrically conductive and highly responsive to electric and magnetic fields.
Plasma is sometimes called the fourth state of matter, and it’s extremely common in space—about 99% of the visible matter in the universe is described as plasma in many scientific summaries.[f]🔗 On Earth, plasma shows up in lightning, neon signs, and many high-energy electrical discharges.
One more useful nuance: plasma is often created by heating a gas a lot, but it can also be produced by other energy inputs (like strong electric fields) that strip electrons away without the “whole gas” needing to behave like an oven-hot flame at all times.[f]🔗 The defining feature isn’t “glowing” or “super hot”—it’s the presence of free charges and how they respond collectively.
Why Temperature and Pressure Both Matter
It’s tempting to picture matter changing state like a simple ladder: add heat and you go solid → liquid → gas → plasma. That’s a good starting mental model, but real materials also care a lot about pressure.[c]🔗 Pressure can push particles closer, making it easier for a liquid to form—or, in some cases, making a solid more stable.
A phase diagram maps the temperature and pressure ranges where solid, liquid, and gas can exist and where they can be in equilibrium.[c]🔗 For water, a famous reference point is the triple point, where all three phases can coexist at once—commonly cited at 273.16 K and about 611.657 Pa.[d]🔗
| State | Shape | Volume | Particle “Feel” | Compressibility | Examples |
|---|---|---|---|---|---|
| Solid | Definite | Definite | Particles vibrate in place; often ordered | Low | Ice, salt, metals, glass (amorphous) |
| Liquid | Takes container shape | Definite | Particles stay close but slide past each other | Low (but not zero) | Water, oils, alcohol |
| Gas | Takes container shape | Fills container | Particles far apart; move freely | High | Air, helium, steam (water vapor) |
| Plasma | Takes container shape | Fills container | Ionized gas: electrons + ions; field-sensitive | High | Neon signs, lightning, auroras, stars |
States of Matter, Mapped by Motion and Pressure
As energy rises, particles move more freely. As pressure rises, particles are pushed closer together. Both knobs matter when a substance decides which state is most stable.
Stability Is a Competition
Thermal motion tries to spread particles out; attractive forces try to keep them together.
Transitions Have “Hidden” Energy
During melting/boiling, energy often goes into rearranging particles rather than raising temperature.
Plasma Adds Charge Effects
Free electrons and ions mean electric and magnetic fields can steer the whole system.
Phase Changes: What Actually Changes During a Transition
A phase change happens when a substance switches between states because temperature and/or pressure cross a boundary where a different arrangement is more stable.[b]🔗 The surprising part is that the temperature can stay flat during the change, because the added energy is used to rearrange particles rather than speed them up.[b]🔗
Think of it like paying for a move: some energy goes into “shipping costs” (breaking and remaking interactions), not into “driving faster.” That’s why melting ice can sit at the same temperature while it absorbs energy.
Common Phase Changes (With Plain Meanings)
- Melting: solid → liquid
- Freezing: liquid → solid
- Vaporization (including boiling): liquid → gas
- Condensation: gas → liquid
- Sublimation: solid → gas (dry ice is a famous example)
- Deposition: gas → solid (frost forming on a cold surface)
- Ionization: gas → plasma (electrons stripped away)
- Recombination: plasma → gas (electrons rejoin ions)
Three Things That Shift During a Phase Change
- Particle arrangement (locked, sliding, free, or charged-and-free)
- Average spacing (usually increases from solid to gas)
- How forces “win” against motion (stronger influence in solids/liquids)
Common Misconceptions and Simple Fixes
Key Terms You’ll See Everywhere
- State of matter
- A practical category for how a substance holds shape/volume and how it flows or compresses.
- Phase
- A region of material with uniform physical properties (like “all liquid water” in a glass).
- Phase diagram
- A map of which phase is stable at different temperatures and pressures.[c]🔗
- Triple point
- A specific temperature and pressure where solid, liquid, and gas can coexist in equilibrium (water’s triple point is widely used as a reference).[d]🔗
- Critical temperature
- The temperature at and above which a substance’s vapor can’t be liquefied by pressure alone.[h]🔗
- Ion / Ionization
- An ion is an atom or molecule with a net charge; ionization is the process that creates charged particles.
- Plasma
- A gas that’s at least partly ionized, containing electrons and ions, and roughly neutral overall.[e]🔗
- Latent heat
- Energy absorbed or released during a phase change without a temperature rise in the material as a whole.[b]🔗
Where the Simple Four-State Model Stops (Limitations)
The “solid, liquid, gas, plasma” set is a great everyday way to classify matter, but it doesn’t describe every extreme or special case. Some materials don’t switch sharply from one state to another in a neat way (glass is a classic example), and under certain conditions substances can form other phases like supercritical fluids where the usual liquid–gas boundary disappears (connected to the critical temperature idea).[h]🔗
Also, many real-world materials are mixtures (foams, gels, emulsions), so asking “what state is this?” can be like asking what genre a song is—usually answerable, but sometimes it depends on which feature you care about most.
FAQ
Answers People Usually Want (Without the Fluff)
Why does adding heat sometimes not raise temperature right away?
During a phase change, energy can be used to rearrange how particles interact (breaking or weakening attractions) rather than increasing particle speed. That’s why melting and boiling can hold steady at a temperature while the change is happening.[b]🔗
Is steam a gas or a vapor?
In everyday speech, “steam” often means the visible mist (tiny liquid droplets). In technical language, water in the gaseous form below its critical temperature is often called a vapor, because it can be condensed by pressure at the same temperature; above the critical temperature, that distinction breaks down.[h]🔗
What’s the simplest definition of plasma?
Plasma is a gas that’s at least partly ionized—meaning it contains free electrons and ions—while being roughly neutral overall. Those charges make it respond strongly to electric and magnetic fields.[e]🔗
Why does pressure matter so much for boiling and condensation?
Pressure changes how hard it is for particles to separate into a gas. Phase diagrams summarize this: different temperatures and pressures favor different phases and define where transitions occur.[c]🔗
Is glass really a solid?
Glass is typically treated as an amorphous solid: it lacks the repeating order of crystals but behaves rigidly at normal timescales. Its structure and formation are closely tied to cooling and atomic mobility.[g]🔗
Do the four states cover everything in the universe?
They cover a lot, but not everything. They’re the most common “big buckets,” and plasma is especially widespread in space. Still, special conditions can produce other phases, and some materials don’t fit neatly into a single label.[f]🔗
Sources
These are high-trust references used to verify definitions, key numbers, and boundary concepts (temperature, pressure, plasma, and phase changes).
- OpenStax – Chemistry 2e: Phases and Classification of Matter (Basic definitions and properties of solids, liquids, gases, and plasma) [a]↩
- OpenStax – Physics: Phase Change and Latent Heat (Why temperature can stay constant during phase changes; latent heat; transition concepts) [b]↩
- Purdue University – Phase Diagrams (How temperature and pressure define phase stability and boundaries) [c]↩
- NIST – Realizing the kelvin: Present realization and dissemination (Reference values tied to the triple point of water used in temperature metrology) [d]↩
- IUPAC Gold Book – Plasma (P04687) (Precise definition: partially ionized gas with electrons and ions; overall neutrality) [e]↩
- U.S. Department of Energy – DOE Explains…Plasma (What plasma is and where it appears; common framing in science communication) [f]↩
- MIT OpenCourseWare – Introduction to Glasses (Amorphous solids: lack long-range order; relationship to cooling and structure) [g]↩
- Purdue University – Critical Temperature and Pressure (Critical temperature concept used to distinguish condensation limits and “vapor” discussions) [h]↩
Once you see states of matter as a balance between particle motion, attraction, and pressure, the whole topic gets easier to predict: solids resist rearranging, liquids rearrange without changing volume much, gases trade cohesion for freedom, and plasma adds the extra twist of charge and fields.