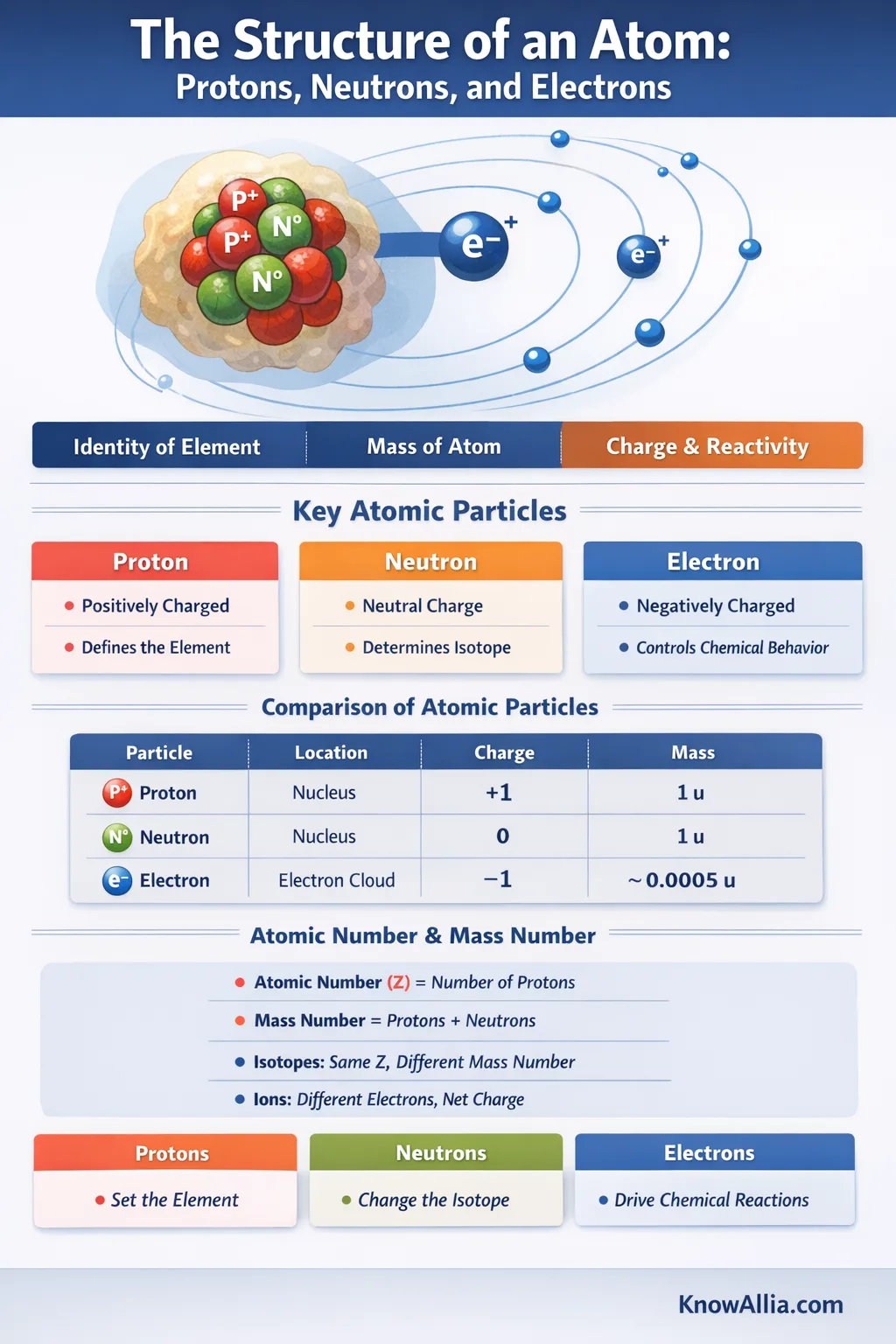
An atom is the smallest particle that still represents a chemical element: a tiny, positively charged nucleus (made of protons and usually neutrons) with electrons around it shaping the atom’s size and behavior.[b]🔗
A Clear Picture Before the Details
Atoms look simple on paper, yet they explain why matter has identity (which element it is), charge (neutral or ion), and patterns (how substances react and bond). Here, we’ll focus on what’s solid and measurable—and we’ll flag what’s inherently “probability-based” in modern physics.
- Protons set the element (atomic number).
- Neutrons help set the isotope (mass number).
- Electrons set the ion’s charge and the atom’s chemical style.
By the end, you’ll know how protons, neutrons, and electrons work together; how to read atomic number and mass number; and why electrons are often described as a cloud instead of neat little tracks.
What an Atom Is
An atom has two main “regions.” The nucleus is compact and positively charged, and it carries almost all of the atom’s mass.[b]🔗 Around it is the electron region, where electrons set the atom’s size and most of its chemical behavior.[b]🔗
If you want one easy mental picture, think of the nucleus as the pit of a peach: small compared with the whole fruit, but it’s where most of the “heft” is concentrated. It’s not a perfect comparison, yet it captures the big idea without pretending atoms are built to scale.
Practical takeaway When people say “this element is different,” they usually mean it has a different number of protons. When they say “this element behaves differently,” they often mean its electrons are arranged differently.
Meet the Three Particles
Proton
A proton lives in the nucleus and carries a positive electric charge. The count of protons is the atomic number, which defines the element.[c]🔗
Neutron
A neutron is also in the nucleus. It has no net electric charge and helps set the nucleus’s total count of “heavy particles,” called the mass number.[d]🔗
Electron
An electron is found outside the nucleus and carries a negative electric charge. Electrons don’t just “sit” somewhere; their locations are described by probabilities and energy states, which is why you’ll often hear about orbitals and configurations.[g]🔗
Mass, Charge, and Where Each Particle Sits
For everyday chemistry and basic physics, three facts do most of the work: protons and neutrons cluster in the nucleus, electrons occupy the surrounding region, and the total charge depends on the balance between protons and electrons.
| Particle | Where It’s Found | Electric Charge | Typical Mass (u) |
|---|---|---|---|
| Proton | Nucleus | +e | 1.007276… u[a]🔗 |
| Neutron | Nucleus | 0 | 1.008664… u[a]🔗 |
| Electron | Electron Region | −e | 0.00054858… u[a]🔗 |
The symbol e is the elementary charge, the fixed charge size used in modern SI definitions.[a]🔗 A proton’s charge is +e, an electron’s is −e, and a neutron’s is 0.
Atomic Number and Mass Number
Two counting ideas show up everywhere in atomic structure:
- Atomic number (Z) = the number of protons in the nucleus.[c]🔗
- Mass number = the total number of protons and neutrons in the nucleus (together called nucleons).[d]🔗
A useful shorthand for charge is: net charge = (number of protons) − (number of electrons). When those two match, the atom is neutral. When they don’t, it’s an ion.[f]🔗
Isotopes and Ions
Isotopes
Isotopes are nuclides with the same atomic number but different mass numbers.[e]🔗 Same element, different neutron count.
- Same protons → same element.
- Different neutrons → different isotope.
- Often very similar chemistry, because chemistry is mainly about electrons.
Ions
An ion is an atomic or molecular particle with a net electric charge.[f]🔗 Ions form when electrons are gained or lost.
- More electrons than protons → negative ion.
- Fewer electrons than protons → positive ion.
- Ion charge changes behavior: bonding, conductivity, and how substances interact.
Electrons: Shells, Orbitals, and Electron Configuration
Electrons are often drawn as dots on rings, but that’s a teaching picture—not a literal track. In modern terms, an electron configuration is the distribution of an atom’s electrons over one-electron wavefunctions called orbitals, following rules such as the Pauli principle.[g]🔗
- Shell
- A broad energy “layer” where electrons can exist. Shells are often labeled by numbers (1, 2, 3…), especially in basic courses.
- Orbital
- A probability-based region tied to a specific energy and shape. Orbitals are a more detailed way of describing where an electron is likely to be found.
- Configuration
- The pattern of how electrons occupy orbitals. This pattern strongly influences an element’s chemistry.[g]🔗
This is why “electron cloud” is a popular phrase: it matches the idea that we can talk about likelihood and energy levels without claiming we know an electron’s exact location like a tiny marble.
How We Know Electrons Have Energy Levels
One strong line of evidence comes from spectroscopy. Atoms and ions produce characteristic spectral lines, and those lines connect directly to energy levels and transitions. NIST’s Atomic Spectra Database organizes critically evaluated data by wavelengths and energy levels, including ground states and ionization energies.[h]🔗
Inside an Atom: What Each Part Controls
The nucleus sets the element’s identity, while electrons shape size, charge, and most chemical behavior.
Protons Define the Element
Change the proton count, and you change what the atom is called on the periodic table.
Neutrons Tune the Isotope
Same element can have different neutron counts, affecting mass and some physical behavior.
Electrons Drive Chemistry
How electrons are arranged across energy levels strongly influences bonding and reactions.
Common Misconceptions and Confusion
Atoms are often taught with simplified drawings. Those drawings help at first, but they can also plant a few sticky misunderstandings.
Confusion
“Electrons orbit the nucleus like planets around the Sun.”
That picture is a starter model. A better idea is probability: orbitals describe where an electron is likely to be found, not a tiny circular track.[g]🔗
Reliable
“Atomic number is the proton count.”
This is the cleanest definition: atomic number is the number of protons in the nucleus.[c]🔗
Confusion
“Mass number is the same thing as atomic weight.”
Mass number is a simple count of protons + neutrons for a specific nucleus.[d]🔗 “Atomic weight” (often called relative atomic mass) is a weighted average across isotopes and depends on natural abundance, so it’s a different idea.
Reliable
“An ion is a particle with net charge.”
This is the official definition used in chemistry terminology: an ion has a net electric charge.[f]🔗
Key Terms (Mini Glossary)
- Atom
- Smallest particle still characterizing an element; nucleus plus electrons.[b]🔗
- Nucleus
- The dense central region containing protons and (typically) neutrons; carries most of the atom’s mass.[b]🔗
- Atomic Number (Z)
- The number of protons in the nucleus.[c]🔗
- Mass Number
- Total number of protons and neutrons (nucleons) in the nucleus.[d]🔗
- Isotopes
- Nuclides with the same atomic number but different mass numbers.[e]🔗
- Ion
- An atomic or molecular particle with a net electric charge.[f]🔗
- Electron Configuration
- The distribution of electrons over orbitals (one-electron wavefunctions), following rules like the Pauli principle.[g]🔗
- Energy Level
- A permitted energy state for electrons in atoms/ions; transitions between levels show up in spectra.[h]🔗
Limitations and What We Still Can’t Point To
Atomic structure is one of the best-tested ideas in science, yet it has built-in boundaries. We can measure masses and fundamental constants to high precision,[a]🔗 and we can map energy levels through spectroscopy data.[h]🔗 But we can’t give a single, always-correct answer to “Where is the electron right now?” because the modern description is probabilistic and depends on what is measured.
Also, many deeper questions—like exactly how nuclear structure emerges from more fundamental particles—go beyond the practical “protons, neutrons, electrons” model. That model remains extremely useful, especially for chemistry and most everyday physics, because it matches what we can reliably predict and observe.
FAQ
Answers People Usually Want First
Are atoms mostly empty space?
In a practical sense, yes. The nucleus holds almost all the mass,[b]🔗 while electrons occupy a much larger region described by probabilities rather than a filled solid. “Empty” doesn’t mean “nothing happens,” though—electric forces and quantum rules are very active there.
What makes one element different from another?
The number of protons. Atomic number is defined as the proton count in the nucleus, and that’s what sets the element’s identity.[c]🔗
Do neutrons change the element?
How does an atom become an ion?
By gaining or losing electrons. An ion is defined as an atomic or molecular particle with a net electric charge.[f]🔗 Changing electron count changes charge without changing the element.
Why do electron configurations matter?
Because electron configuration describes how electrons are distributed across orbitals, and that distribution strongly influences bonding and reactions.[g]🔗
How do we know atoms have specific energy levels?
Atoms and ions produce characteristic spectra. NIST organizes and evaluates data on atomic energy levels, wavelengths, and transition probabilities in its Atomic Spectra Database.[h]🔗
Sources
These references were chosen for strong editorial standards and stable, citable definitions. Use them if you want to verify terminology or numerical values.
- NIST – CODATA Recommended Values of the Fundamental Physical Constants: 2022 (NIST SP 961) (particle masses, elementary charge, and related constants) [a]🔗
- IUPAC Gold Book – “Atom” (A00493) (definition of atom; nucleus mass dominance; electrons shaping size) [b]🔗
- IUPAC Gold Book – “Atomic Number” (A00499) (atomic number as proton count) [c]🔗
- IUPAC Gold Book – “Mass Number” (M03726) (mass number as total nucleons) [d]🔗
- IUPAC Gold Book – “Isotopes” (I03331) (same atomic number, different mass numbers) [e]🔗
- IUPAC Gold Book – “Ion” (I03158) (definition of ion as a net-charged particle) [f]🔗
- IUPAC Gold Book – “Configuration of Electrons” (C01248) (definition of electron configuration and orbitals) [g]🔗
- NIST – Atomic Spectra Database (SRD 78) (how spectroscopy data is organized by wavelengths, energy levels, and transitions) [h]🔗
- Encyclopaedia Britannica – “Atom” (historical models and broad overview of atomic structure concepts) [i]🔗