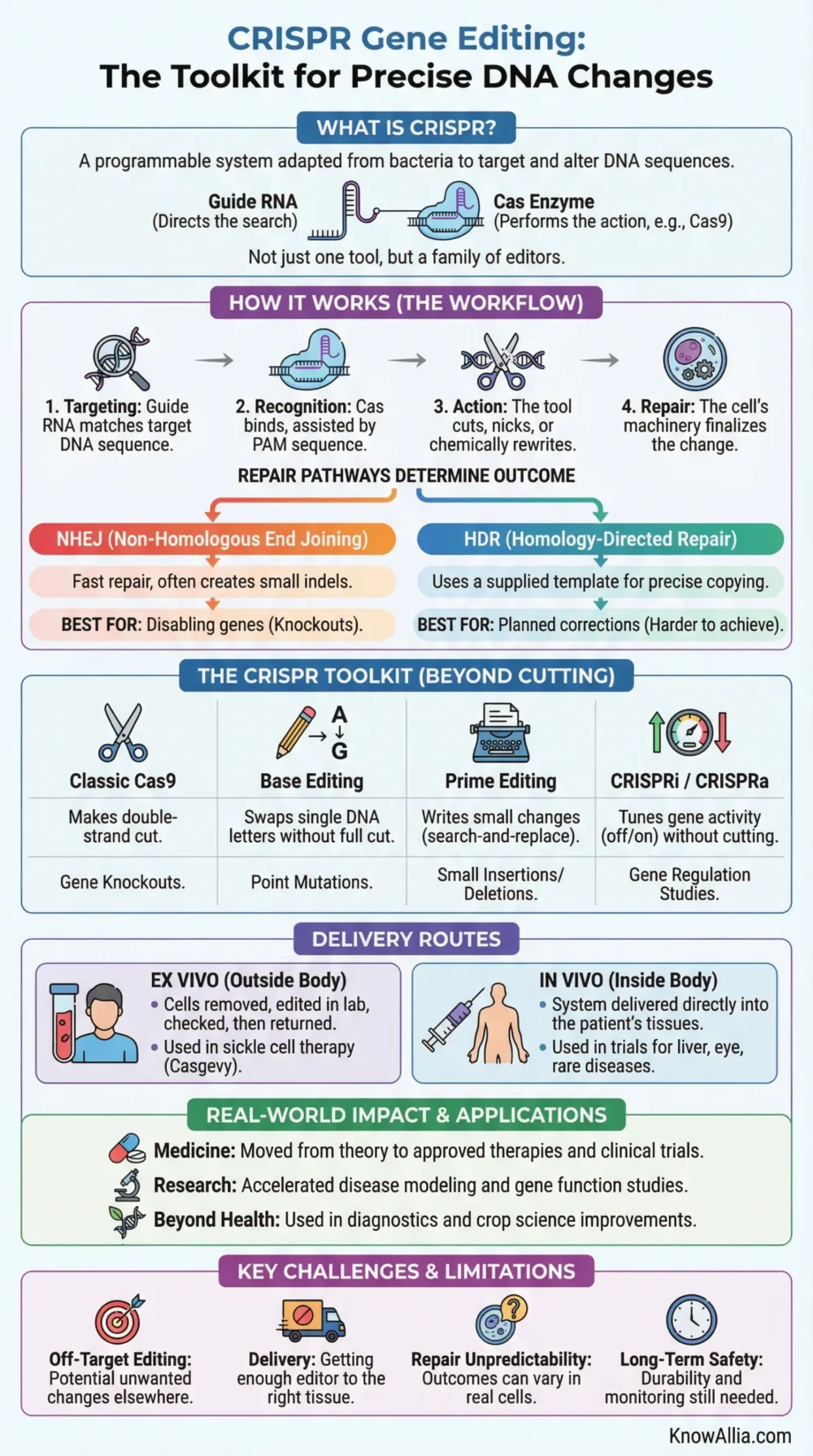
CRISPR gene editing is a way to target a chosen DNA sequence and cut, rewrite, silence, or tune it with unusual precision. The method used in labs today was adapted from a bacterial defense system, and the most familiar version pairs a guide RNA with a Cas enzyme such as Cas9 to act on a selected spot in the genome.[a]
A Clear Starting Point
CRISPR is not one single action. It is a toolkit. In one version, it cuts DNA and lets the cell repair the break. In newer versions, it can swap one DNA letter for another or write a small new sequence without making the same kind of full double-strand cut.
That is why CRISPR matters: it changed how researchers study genes, how some cell therapies are built, and how scientists think about treating inherited conditions. It also changed the pace of lab work, because designing a new target is often much easier than with older editing systems.
- Classic Cas9 editing often depends on the cell’s own repair machinery.
- Base editing and prime editing aim for more exact changes in many settings.
- Some medical uses are already real care, while many others are still in trials.
You will see where the edit really happens, why the cell’s repair response matters as much as the cut itself, how base editing and prime editing differ from standard Cas9 work, and where approved treatment ends and active research begins.
What CRISPR Is
CRISPR stands for “clustered regularly interspaced short palindromic repeats.” In bacteria, these repeated DNA segments are part of an immune defense that helps recognize and cut genetic material from invading viruses. Researchers learned how to simplify that system for lab use, and the work behind CRISPR-Cas9 was recognized with the 2020 Nobel Prize in Chemistry.[b]
- Programmable target site
- Guide RNA directs the search
- Cas enzyme performs the action
- Cell repair shapes the result
That last point is often underexplained. The editor can aim at the DNA address, but the final outcome depends on what the cell does next. A clean-looking design on paper can still lead to different repair outcomes in real cells, which is why editing is both a molecular tool and a cell biology problem.
How Editing Works
For the widely used CRISPR-Cas9 setup, the workflow is fairly simple on the surface. A guide RNA is designed to match the target DNA sequence, the Cas9 protein binds with that guide, and the complex searches the genome for a matching site next to a short sequence called a PAM. Once it finds the right place, Cas9 cuts the DNA, and the cell repairs the break.[c]
- Targeting: a guide RNA is built to match the DNA region scientists want to affect.
- Recognition: the CRISPR complex scans DNA and locks onto the matching sequence.
- Action: the Cas enzyme cuts DNA, nicks one strand, or carries a chemical editing module, depending on the tool.
- Repair or rewriting: the cell’s own machinery completes the change.
A useful analogy is to picture classic Cas9 as marking one sentence in a long printed book and cutting that exact line. The cut is precise, but the final wording depends on how the repair crew tapes the page back together. If the cell rejoins the DNA quickly, a few letters may be lost or added. If a repair template is available, a more planned sequence can sometimes be copied in.
When the Cell Uses NHEJ
Non-homologous end joining is the fast repair route. It often creates small insertions or deletions, called indels. That makes it useful for disabling a gene.
When the Cell Uses HDR
Homology-directed repair can copy a supplied template into the cut site. That makes more exact replacement possible, but it is often harder to achieve in many cells and tissues.
The turning point in understanding CRISPR: the edit is not finished when DNA is targeted. In many experiments, repair biology is what decides whether the result becomes a knockout, a correction, a mixed outcome, or no useful change at all.
Editing Tools in Plain Language
Many articles still talk as if CRISPR means one thing. It does not. Today, the word usually refers to a family of programmable tools. Some cut DNA. Some rewrite single letters. Some do not cut DNA at all and instead tune gene activity.[d][e][h]
| Approach | What It Does | Best Fit | Main Limitation |
|---|---|---|---|
| Classic Cas9 | Makes a double-strand cut at a selected DNA site. | Gene knockout, target validation, some cell therapy workflows. | The cell’s repair outcome can vary from one edit to another. |
| Base Editing | Changes one DNA letter to another without the same kind of full double-strand cut. | Point mutations where a single-letter swap may help. | Only certain letter changes are possible with a given editor. |
| Prime Editing | Uses a nickase plus reverse transcriptase to write a planned small change. | Small substitutions, insertions, and deletions. | Design and delivery are more complex, and efficiency can vary by cell type. |
| CRISPRi / CRISPRa | Uses a non-cutting CRISPR system to turn gene activity down or up. | Gene function studies and cell-state control. | It changes expression, not the DNA sequence itself. |
How a CRISPR Edit Turns Into a Real DNA Outcome
Targeting is only the start. The final result depends on the editing tool and the cell’s own response.
A guide RNA is designed to match the target DNA sequence.
The Cas system binds the matching region, usually with help from a PAM.
The tool cuts, nicks, or chemically rewrites a base depending on the editor.
Repair pathways or reverse-transcriptase copying shape the final sequence.
NHEJ
Fast repair that often leaves small insertions or deletions. Useful for turning genes off.
HDR
Template-based repair that can support a planned correction, though it is not equally efficient in all cells.
Base and Prime Editing
Built for finer DNA changes, often with less reliance on a full double-strand break.
What CRISPR Changes
The first change was in research speed. CRISPR made it easier to knock out genes, build disease models, run pooled screens, and ask what happens when a gene is turned down, turned up, or subtly rewritten. That shifted everyday lab work from “Can we edit this?” to “Which editing strategy fits this question best?”[f]
In medicine, the clearest change is that gene editing moved from theory to approved care. In December 2023, the U.S. FDA approved Casgevy, the first FDA-approved therapy that uses CRISPR/Cas9. It is an ex vivo treatment for certain patients with sickle cell disease, meaning blood-forming stem cells are edited outside the body and then returned to the patient.[g]
CRISPR also changed what seems possible in rare disease care. Human studies have already tested in vivo editing in the liver, and in 2025 researchers reported a personalized gene-editing treatment for an infant with severe CPS1 deficiency. Those advances matter, but they do not mean every genetic condition is now editable in a simple way. Most uses are still in research or clinical trials.[i][j]
Where the Biggest Changes Show Up
- Basic biology: cleaner gene-function studies and faster model building.
- Cell therapy: edited cells can be checked before they go back into the patient.
- Rare disease medicine: single-gene conditions are often the first place where editing logic is easiest to define.
- Diagnostics: some CRISPR systems can detect nucleic acids, not just edit them.[k]
- Crop science: plant researchers use CRISPR to study and improve traits with more targeted genetic changes than older breeding-only approaches.[l]
How the Editor Reaches Cells
A common gap in popular explanations is delivery. Designing a guide RNA is only part of the job. The harder question is often how to get the editor into the right cells, in the right amount, at the right time.
Ex Vivo Editing
Cells are taken out of the body, edited in the lab, checked, and then infused back. This route gives researchers more control over which cells were edited and how well the process worked. It is the route used in the first FDA-approved CRISPR therapy for sickle cell disease.
In Vivo Editing
The editing system is delivered directly into the body, often with a delivery vehicle such as lipid nanoparticles or a viral vector. This is attractive for tissues that cannot be removed and returned, but it raises tougher questions about targeting, dose, and long-term follow-up.
That is one reason blood disorders became early leaders for clinical success. Blood-forming stem cells can be collected, edited outside the body, and measured before reinfusion. Many other tissues do not offer that same level of access.[m]
Common Points of Confusion
In practice, it names a broader programmable system that now includes DNA-cutting editors, base editors, prime editors, and non-cutting gene-regulation tools.
Many edits do not add anything. They may disable a gene, reactivate a fetal program, or change one DNA letter.
The mutation may be simple, but the biology may not be. Scientists still have to reach the right cells, edit enough of them, and avoid unwanted changes.
The field already has approved therapy and strong clinical momentum, yet most uses are still being tested, refined, and followed over time.
Limits and Open Questions
CRISPR is powerful, but it is not a guarantee of a clean, permanent, universal fix. The best-known technical concern is off-target editing, where the editor acts at a DNA site that looks similar to the intended target. Regulators also care about long-term safety monitoring, especially in therapies built around permanent DNA change.[p]
- Targeting accuracy: guide design has improved, but unwanted edits still need careful testing.
- Delivery: getting enough editor into the right tissue is still one of the field’s hardest tasks.
- Repair unpredictability: a targeted cut can lead to more than one outcome inside real cells.
- Durability: some treatments may last for years, but long-term monitoring is still needed in many settings.
- Scale: personalized editing for very rare cases is now possible in principle, yet making that fast and broadly accessible remains a practical challenge.
What We Still Do Not Know
Scientists do not yet know the best editing format for every tissue, how widely ultra-personalized treatments can be scaled, or which long-term effects may matter most across different delivery methods. Global health bodies also stress that human genome editing needs strong oversight around safety, effectiveness, transparency, and follow-up.[q]
Key Terms in Plain English
- Guide RNA
- The RNA sequence that tells the CRISPR system where to go in the genome.
- Cas9
- A CRISPR-associated enzyme that can cut DNA at the selected site.
- PAM
- A short DNA motif next to the target that helps enzymes such as Cas9 recognize where action is allowed.
- NHEJ
- A fast DNA repair pathway that often leaves small insertions or deletions.
- HDR
- A repair pathway that can copy in a planned DNA template.
- Base Editing
- A way to swap one DNA letter for another without relying on the same full double-strand break used in classic Cas9 editing.
- Prime Editing
- A method that uses a nickase and reverse transcriptase to write a planned small DNA change.
- CRISPRi / CRISPRa
- Non-cutting CRISPR tools that turn gene activity down or up.
- Ex Vivo
- Editing cells outside the body and returning them afterward.
- In Vivo
- Delivering the editing system directly into the body.
- Off-Target Edit
- An unintended change at a DNA site other than the main intended target.
Put together, these terms explain why CRISPR is better understood as a programmable gene-control and gene-change platform, not just a pair of molecular scissors. What changes in practice depends on the editor chosen, the cell type, the delivery route, and the biology of the disease or trait being studied.
FAQ
Questions Readers Usually Ask
What is the difference between CRISPR and CRISPR-Cas9?
CRISPR is the wider system and tool family. CRISPR-Cas9 is one well-known version that uses the Cas9 enzyme to target and cut DNA.
Does CRISPR always cut DNA?
No. Classic Cas9 usually cuts DNA, but base editing, prime editing, CRISPRi, and CRISPRa were developed to make other kinds of changes or to regulate genes without the same type of full double-strand cut.
Why was sickle cell disease an early clinical success for CRISPR?
Its treatment can use ex vivo editing of blood-forming stem cells, which are easier to collect, edit, test, and return than many cells in solid organs.
Can CRISPR add a new gene?
It can support sequence insertion in some settings, but many real edits simply disable a gene, restore a regulatory pattern, or rewrite a small number of DNA letters.
What is the difference between ex vivo and in vivo editing?
Ex vivo editing happens outside the body and the edited cells are returned later. In vivo editing delivers the editing system directly into the body so it acts inside the patient’s tissues.
Is CRISPR only used in medicine?
No. It is also used in basic research, crop science, and some diagnostic systems that detect genetic material rather than edit it.
Sources
- [a] NHGRI – CRISPR — Definition, origin, and lab adaptation.
- [b] Nobel Prize – The Nobel Prize in Chemistry 2020 Press Release — Nobel recognition for the development of CRISPR-Cas9 genome editing.
- [c] NHGRI – How Does Genome Editing Work? — Targeting, PAM recognition, cutting, and repair pathways.
- [d] Nature – Programmable Editing of a Target Base in Genomic DNA Without Double-Stranded DNA Cleavage — Original base-editing paper.
- [e] Nature – Search-and-Replace Genome Editing Without Double-Strand Breaks or Donor DNA — Original prime-editing paper.
- [f] Broad Institute – Genetic Perturbation Platform — CRISPR editing, CRISPRi, and CRISPRa in research workflows.
- [g] FDA – FDA Approves First Gene Therapies to Treat Patients with Sickle Cell Disease — First FDA-approved CRISPR-based therapy and its clinical context.
- [i] NEJM – CRISPR-Cas9 In Vivo Gene Editing for Transthyretin Amyloidosis — Early human in vivo editing example.
- [j] NIH – Infant With Rare, Incurable Disease Is First to Successfully Receive Personalized Gene Therapy Treatment — Personalized gene editing reported in 2025.
- [k] Nature Biomedical Engineering – CRISPR-Based Diagnostics — Detection uses of CRISPR systems.
- [l] USDA National Agricultural Library – Crop Genetic Enhancement Through Genome Editing Technology and Future Agriculture Leadership — Crop research applications of genome editing.
- [m] NCBI Bookshelf – CRISPR Technologies for In Vivo and Ex Vivo Gene Editing — Clear explanation of ex vivo and in vivo delivery routes.
- [n] Nature – Programmable Editing of a Target Base in Genomic DNA Without Double-Stranded DNA Cleavage — Why base editing differs from classic DNA cutting.
- [o] Nature – Search-and-Replace Genome Editing Without Double-Strand Breaks or Donor DNA — Why prime editing differs from classic DNA cutting.
- [p] FDA – Summary Basis for Regulatory Action for CASGEVY — Off-target editing and long-term safety follow-up.
- [q] WHO – Human Genome Editing: Recommendations — Safety, oversight, transparency, and follow-up in human genome editing.